Table of Contents
Introduction of pyridine
Introduction of pyridine, a heterocyclic compound with a six-membered ring containing five carbon atoms and one nitrogen atom, holds a prominent place in the field of organic chemistry. Its unique structure and diverse properties have made it an essential building block for numerous pharmaceuticals, agrochemicals, and other chemical applications. Pyridine has found extensive use as a solvent, catalyst, and intermediate in various chemical reactions.
Structure of pyridine
Pyridine is an unsaturated 6 membered heterocyclic ring consist of nitrogen as hetero atom. It passes planar conjugated ring structure consists of six delocalized π electrons. The aromatic nature arises from the three double bonds present in the ring as six delocalized π electrons. So, it follows Huckel’s rule hence it is aromatic compound.

Hybridization of pyridine
Pyridine’s nitrogen is in Sp² orbital, while the nitrogen the aliphatic amines is in Dp³. Due to more ‘s’ character, the Sp² orbital are however smaller than the Sp³ orbitals. Thus, the lone pair of electrons in the nitrogen of pyridine is closely associated with the nitrogen nucleus indicating that the lone pair of electron is less available for the formation of bond with proton. This reduces the relative basicity.
Basicity of pyridine
Pyridine is a very strong base due to the presence of a negatively charged nitrogen atom. The basic nature is also imparted because of the lone pair undergoes rapid protonation. However, its basicity is much less than in actual than expected. Pyridine is a tertiary amine, it’s a basicity in aqueous solution is lesser when compared to that of the tertiary aliphatic amines and ammonia.
Dipole moment of pyridine
Dipole moment governs that charge separation in pyridine: pyridine(1.17D) and pyrrole(1.81D) have a large dipole moment than pyridine. Dipole moment in all the case directs towards the N-atom. In Sp² hybridised orbital of pyridine the electronegative of nitrogen is more than in Sp³ orbital of triethyl(amine). This increased electronegativity tightly bonds the lone pair of electrons and prevents their protonation.
Pyridine’s basicity is due to the presence of Sp² hybridised lone pair of electrons in its N-atom. The basic nature of pyridine also indicates that these electrons are not involved in the formation of delocalised π-molecular orbital. The lone pair of electrons, however readily forms a new N-H bond with proton.
Pyridine is more basic than pyrrole
The basicity of pyrrole is less than pyridine because the lone pair of electron N-atom of pyrrole is in the p-orbital & forms a part of the delocalised π-molecular orbital, also these electrons don’t form a new N-H bond with proton.
Physical properties of pyridine
1. It is a colourless liquid.
2. The boiling point of pyridine is 115°C.
3. It has a disgusting odour.
4. It is miscible with water & most organic solvents.
5. It forms an azeotrope with water, which boils at 92-93°C.
Synthesis of pyridine
1. Hantzsch pyridine synthesis
Condensation of 2 moles of B-dicarbonyl compounds/B-keto esters with aldehyde & ammonia.
2. From Diel’s-Alder cycloaddition
Diel’s-Alder cycloaddition of oxazole and dienophile produce pyridine derivatives.
3. From 1,5-diketones
1,5-dicarbonyl compounds on reaction with ammonia gives 1,4-dihydropyridines, which are easily dehydrogenated to pyridine.
4. Guareschi pyridine synthesis
The preparation of pyridine derivatives by condensation of acetoacetic esters and cyanoacetamide.
Chemical reactions of pyridine
1. Nitration
2. Sulphonation
3. Bromination
4. Reduction
Medicinal uses of pyridine
1. It is used as a starting material in the preparation of the sulphapyridine, zelatin.
2. It is used as a precures or to agrochemical & pharmaceutical.
3. The pyridine ring occurs in many important compounds including azamines & the vitamins & pyridoxine.
4. Isoniazid is an antibiotic used for the treatment of tuberculosis contains pyridine rings.
5. Pyridine is added to ethanol to make it unsuitable for drinking.
6. It has been used to prevent or treat a certain nerve disorder caused by certain medications.

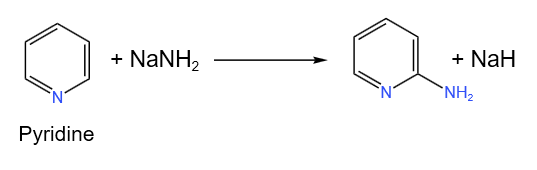

Dear mimprovement.com admin, Good to see your posts!
Hi mimprovement.com administrator, Thanks for the educational content!