Complexometric titration
A complexometric titration is a type of titration that is used to determine the concentration of an analyte in a solution. It is also called an analytical titration.
The analyte being tested will have a ligand that binds to the analyte, and this ligand will be dissolved in the solution being tested. The titrant will then be added to the solution and titrated until all of the ligand has been consumed by the analyte. This process can be done either manually or with a volumetric pump.
Complexometric titration is a way of determining the concentration of an analyte in a solution. The titrant is added to the solution, and the end point is detected by measuring the pH. The end point is detected by measuring the pH. The end point can be determined either by using a pH meter or by using indicator solutions, such as phenolphthalein or methyl red, that change color at different pH values.
So let us now understand what happens inside this titration, When the complexing agent from your burette arrives in the conical flask So the metal ions present in your conical flask form the co-valent vent with the complexing agent.
So now we talk about what are the complexing agents used in this titration. EDTA(Ethyline diamine tetra acetic acid), EDTA is a multi-dented complexing agent.
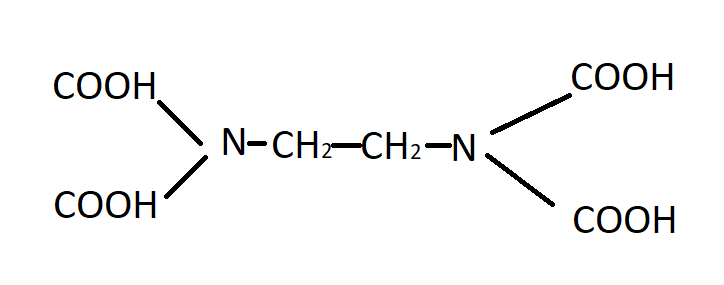
Hard water
Such water is called hard water in which there is an abundance of mineral salts. In this, carbonates of calcium and magnesium (CaCO3, MgCO3) and bicarbonates (Ca (HCO3)2, Mg (HCO3)2) are present. Its simple identity is that it does not produce froth with soap.
If there is a deficiency of magnesium in our body, then our growth slows down, our kidneys are affected and our hair starts falling. Therefore, the consumption of hard water is beneficial for health. Yes, it definitely makes our skin dry and it also damages our hair.
Complexometric titration, further notes will be uploaded very soon


To the mimprovement.com administrator, Your posts are always well presented.